what substances are thought to be main sources of chlorine radicals in the atmosphere
Dudley Shallcross and Tim Harrison explain how a quantum has allowed us to written report Criegee biradicals, and what this could mean for atmospheric scientific discipline

A family of Criegee biradicals, an atmospheric speciesstart postulated in 1949, has recently been detected by an international team of chemists in the US and UK. Criegee biradicals, as their proper name suggests, can be thought of equally having two unpaired electrons on different atoms. These highly reactive – and therefore short-lived – species are formed when volatile unsaturated organic compounds react with ozone. Their discovery may modify the way we think about atmospheric chemistry and sheds further light on the mechanism of ozonolysis.
Gratuitous radicals are very of import in many chemical systems. They can be generated by homolytic cleavage of a covalent bond, where the shared electron pair separates evenly, with one electron going to each of the two molecular fragments. A post-16 level example would be cleaving molecular chlorine to form chlorine radicals, Cl•:
Cl2 → Cl• + Cl•
The dot (•) is used to represent the unpaired electron. This reaction is called an initiation reaction and is endothermic – information technology requires free energy to break the bond. This energy could come from heating Cl2 in the gas phase, or from irradiating the gas with light. In this example, the wavelength is in the region of 350 nm. Once formed, the radical tin can take part in propagation reactions.
In propagation steps, a radical is consumed simply at least one new radical is formed, allowing the reaction to keep. If methane (CH4) is present in the gas mixture with the irradiated chlorine, several possible propagation reactions can occur. Most of these involve radical intermediates abstracting hydrogen atoms from methane derivatives to make HCl gas and new carbon-centred radicals, or reacting with Cl2 to generate more chlorine radicals. A choice of the possibilities is shown beneath:
Cl• + CH4 → CH3• + HCl
CHthree• + Cl2 → Cl• + CH3Cl
Cl• + CH3Cl → CH2Cl• + HCl
CH2Cl• + Clii → Cl• + CHiiCl2
Therefore, consecutive chlorination can occur until tetrachloromethane (CCl4) is formed. Nonetheless, these reactions practise not propagate endlessly. Eventually, a termination reaction occurs, in which ii radicals combine to form a neutral molecule, for instance:
CHthree• + CHthree• → CiiH6
Cl• + CHiii• → CH3Cl
Cl• + Cl• → Cl2
Radical changes
Gratis radical chemistry is very important in the Earth's atmosphere. It regulates ozone levels in the stratospheric 'ozone layer' – and leads tosevere ozone depletionin polar regions each bound. Lower down, in the troposphere (the everyman x km of the atmosphere) free radicals also drive photochemical smog formation. If a gratuitous radical with one unpaired electron reacts very quickly with other molecules, we would expect a biradical, with two unpaired electrons, to react very quickly indeed.
Free radicals regulate ozone levels in the stratosphere
Plants release a wide range of organic molecules, generally termed volatile organic compounds (VOCs), into the atmosphere. We know that some attract pollinators, and some are released when the institute is wounded to concenter predators of any is eating the found. But there are many for which we take no explanation.
Among these VOCs are alkenes, with carbon-carbon double bonds. Ozone (O3) reacts with alkenes, and the reaction charge per unit depends on the alkene's structure. The rate is accelerated when there are more alkyl groups around the double bond. For example, the relative ozonolysis rates of ethene, 2-methylpropene, 2-methyl-2-butene and 2,3-dimethyl-2-butene is in the ratio 2:15:500:1250.
Rudolf Criegee and Gotthilf Wenner first proposed in 1949 that ozonolysis of alkenes proceeds through carbonyl oxide intermediates, such every bit •CH2OO• in the case of ethene. Ozone adds across a double bond to produce a cyclic primary ozonide, which speedily rearranges to a secondary ozonide, then decomposes to produce a carbonyl and a biradical carbonyl oxide, known as a Criegee intermediate.
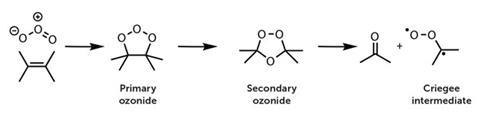
Until recently, no ane had detected Criegee biradicals in the gas phase for two reasons. First, they react extremely quickly. Secondly, alkene ozonolysis reactions - which were thought to be the only way to make them - are too slow in laboratory systems to generate enough Criegee biradicals to detect.
Two things coincided to make Criegee biradical detection possible. Firstly, a fast set of reactions were shown to produce the showtime Criegee biradical, •CH2OO•. Secondly, tuneable, high intensity beams of light generated at a synchrotron facility (the Advanced Calorie-free Source at the University of California in Berkeley, US), allowed •CH2OO• to be detected by photoionisation mass spectrometry (PIMS).
CHtwoI2+ hv → CH2I• + I• (λ ~260 nm)
CH2I• + O2 → •CH2OO• + I•
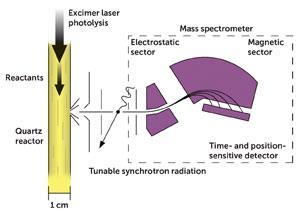
All the required gases flow down a quartz reactor. A light amplification by stimulated emission of radiation pulse is so fired into the quartz tube to initiate the reactions that form the Criegee biradical. On i side of the quartz reactor is a pinhole through which some of the gas mixture can menses. This gas mixture is and then exposed to high energy light from the synchrotron. This will ionise •CHtwoOO• (and all other species that can exist ionised at these energies) into a positive ion. Using electrostatic plates and a magnetic sector, all the ions can be directed to a fourth dimension- and position-sensitive detector to generate a mass spectrum. Nosotros tin follow the meridian corresponding to the Criegee biradical at 46.005 mass units, and watch information technology decay over time.
By adding co-reactants, such as NO2, to the initial gas mixture, we run into that the rate of decay increases. From this nosotros can work out how fast •CH2OO• is reacting with the co-reactant. This method also works for generating •CH(CH3)OO• by photolysis of CH3CHI2•. Both these Criegee biradicals (•CH2OO• and •CH(CH3)OO•) seem to react especially quickly with SO2, NO2 and CF3COCF3•. They probably form initial adducts, which and so decompose to form products.1,2,3. Previous indirect studies suggested that the reactions of Criegee intermediates were much slower – this shows the value of direct measurements.
Into the atmosphere
Criegee biradicals react very quickly with And so2 and NO2 to form And sothree and probably NO3 (although this is all the same being verified):
•CH2OO• + Soii → CH2O + SOiii
•CH2OO• + NO2 → CH2O + NO3
SO3 and NO3 are very reactive in the atmosphere and can go on to grade sulfuric acid, nitric acid and organic nitrates. All of these compounds will add to the aerosol level in the atmosphere, either by adding to existing aerosol particles or, in the case of sulfuric acid, interim equally a site for new droplets particle germination. Sulfuric acrid is extremely hygroscopic – water sticks to it very hands. This process can seed new aerosol formation as the initial cluster of sulfuric acrid and water molecules grows into bigger particles.
In urban areas we would want to avoid high levels of aerosols as they are associated with poor wellness and poor air quality. Still, aerosol particles (not-soot ones, at least) tend to cool the planet down and contribute to a phenomenon known as global dimming or global cooling. The aerosol particles can act like fiddling mirrors and reflect visible calorie-free from the Sun back to infinite. This reflection cools the planet. Well-nigh 24% of the free energy from the sun is reflected by clouds and aerosols, with a further vi% reflecting off icy surfaces like Antarctica, leading to a cooling effect of well-nigh 25°C.4,5 This means in that location is a lot of interest in Criegee biradicals because they may exist important in climate cooling.
The hunt continues

Where would high concentrations of Criegee biradicals be plant? Where at that place are high concentrations of alkenes and ozone. That would mean inside or downwind of urban areas, as well as over forests. Downwind of urban areas, particularly on sunny days, loftier concentrations of ozone form role of photochemical smog. Vehicle emissions volition release alkenes into the temper besides. In these polluted plumes, it is perfectly possible to generate loftier ozone and high alkene concentrations, leading to elevated concentrations of Criegee biradicals.
Forests are rich sources of a range of volatile organic molecules, many of which are unsaturated and therefore concentrations of alkenes in and above a forest canopy are large. Ozone levels increase with altitude, so when atmospheric currents bring air down from higher altitudes to a woods environs, high concentrations of ozone can one time once more see high concentrations of alkenes leading to loftier Criegee biradical concentrations. VOC emissions from plants will increase with rising surface temperature, and so as the climate warms and surface temperatures rise nosotros may wait Criegee biradicals to become fifty-fifty more important.
Source: https://edu.rsc.org/feature/radical-changes-in-our-atmosphere/2000107.article
0 Response to "what substances are thought to be main sources of chlorine radicals in the atmosphere"
Post a Comment